Published on
Updated
Reading 2 mins.
in collaboration with
Dr. David Picovski (Plastic surgeon)
Medical validation:
January 28, 2023
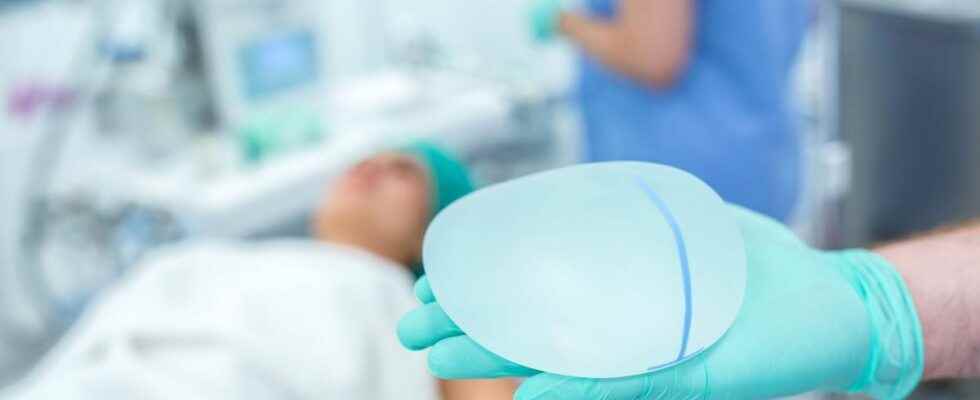
Faced with previous scandals related to the fitting of PIP breast prostheses, the ANSM has stepped up surveillance of the implant market in France. Controls, which make it possible to avoid the marketing of dubious implants but also to protect women from possible undesirable effects.
Infections, skin necrosis, cancers, pain… To avoid the occurrence of adverse effects in patients who have had breast implantation, the National Agency for the Safety of Medicines and Health Products (ANSM) closely monitors the implementation of the prosthetics market. It has published new data in this regard.
A systematic analysis of the surface type of implants
Since the ban on so-called “macro-textured” breast implants (whose grainy surface has been designed to better adhere to tissue) and those coated with polyurethane – accused of being the cause of a rare cancer of the lymph nodes (ALGC- AIM) – ANSM proceeds “a systematic analysis of the type of surface of the implants as soon as they are notified of marketing on the French market, in conjunction with the European Membrane Institute“.
“This control makes it possible to verify that no new implant marketed has a texture similar to those which were banned in 2019.“, specifies the ANSM in a press release dated January 26, 2023.
103 AIM-ALCL cancer cases were detected
Another checkpoint: the number of cases of anaplastic large cell lymphoma (ALCL-AIM), this famous extremely rare cancer associated with the placement of breast implants.
If, on January 17, 2023, the ANSM recorded 103 cases of ALCL associated with the wearing of implants, only a detailed analysis of the first 89 cases could be made – from 2011 to the end of 2022.
Of these cases, 89 patients were diagnosed with anaplastic large cell lymphoma with breast localization, of which 3 cases were bilateral breast localization.
“The number of diagnosed cases peaked in 2019 (year of the health policy decision prohibiting certain families of implants) certainly due to increased detection of BIA-ALCL cases. Since 2019, a decrease in detections has been observed but should be taken with caution due to the impact of the Covid crisis“, specifies the press release.
The Agency also specifies that out of all the data collected, there is no element indicating “an increased risk of occurrence of LACG-AIM for implants currently marketed in France“.
A low risk, which does not surprise Dr. Picovski, cosmetic and plastic surgeon.
“Even if zero risk does not exist in aesthetic medicine, as soon as a prosthesis with a smooth envelope (and not textured) is placed, the risks of anaplastic large cell lymphoma drop. The low number of cases, revealed here by the ANSM, speak for themselves“.
Finally, for patients with the ASIA syndrome – described in women wearing silicone breast implants and characterized by fatigue, loss of memory, skin rash or even joint pain – the Agency specifies that the data to be this subject in France are insufficient to draw conclusions.